Master Breakthrough Therapy Compliance Strategies for Success

Introduction
In the fast-paced world of pharmaceuticals, the FDA's designation of breakthrough therapy serves as a beacon of hope for patients confronting severe conditions. This designation not only accelerates development and review processes but also cultivates innovation, compelling pharmaceutical companies to explore new frontiers in treatment options.
However, the complexities of compliance with breakthrough therapy regulations present formidable challenges that can significantly impact a company's success. How can organizations effectively implement compliance strategies to not only meet regulatory expectations but also enhance patient access to life-saving therapies? By navigating these challenges with strategic foresight, companies can position themselves to thrive in this dynamic landscape.
Define Breakthrough Therapy and Its Importance in Pharmaceuticals
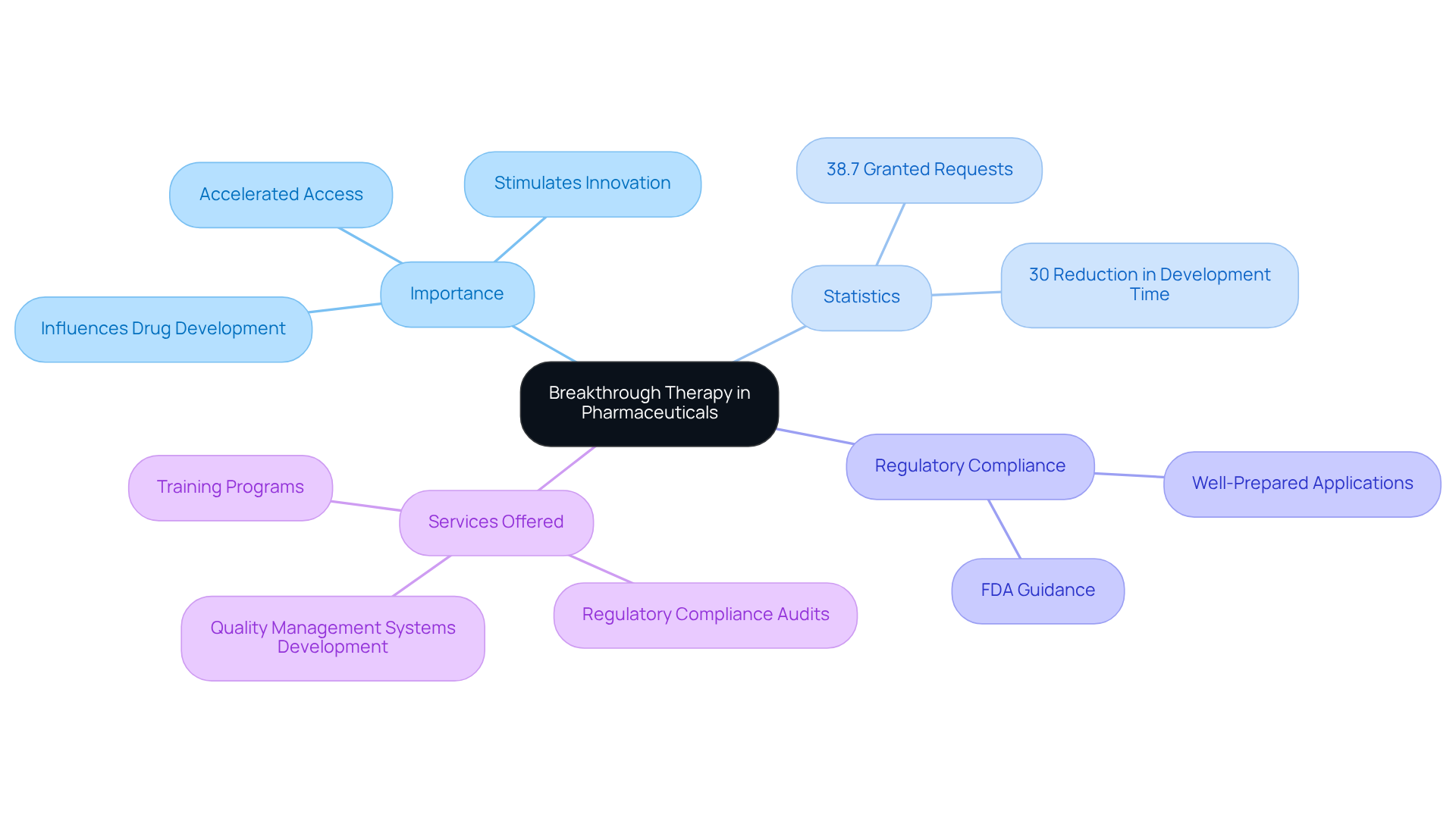
The FDA awards the designation of breakthrough therapy to medications and biologics that show significant advancements over existing treatments for severe or life-threatening conditions. This designation as a breakthrough therapy is crucial because it accelerates both development and review processes, allowing patients to gain quicker access to potentially life-saving treatments.
Beyond expedited timelines, this stimulates innovation in drug development, encouraging pharmaceutical firms to invest in research for conditions with limited treatment options. As of mid-2024, approximately 38.7% of requests for breakthrough therapy designation have been granted, highlighting the importance of .
By grasping the nuances of this designation, stakeholders can effectively align their adherence strategies with regulatory expectations, ensuring they meet the criteria for designation and subsequent approval.
, including:
All tailored to assist clients in associated with innovative treatments. The FDA's commitment to addressing requests within 60 days further emphasizes the significance of this program in enhancing patient access to effective treatments.
Furthermore, breakthrough therapy has been shown to by 30%, significantly influencing the landscape of pharmaceutical innovation. Understanding these dynamics is essential for stakeholders aiming to .
Explore Regulatory Framework for Breakthrough Therapy Designation
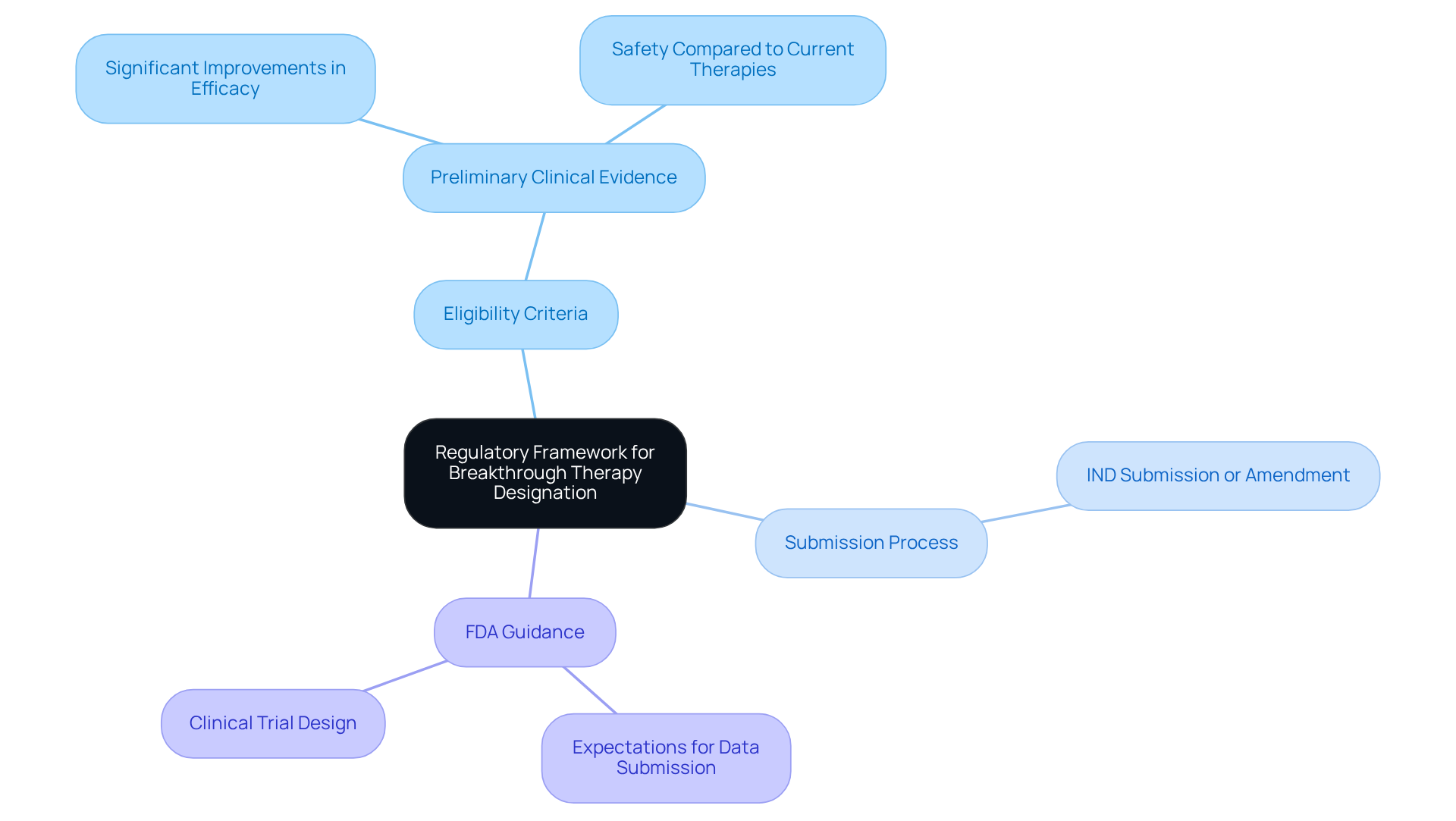
The for is primarily established by the FDA's Safety and Innovation Act. To qualify as a breakthrough therapy, a treatment must target a serious condition and show over existing options. The FDA promotes early and frequent communication between sponsors and regulatory bodies, which is essential for a . Key components of this framework include:
- Eligibility Criteria: The therapy must present preliminary clinical evidence indicating compared to current therapies.
- Submission Process: Sponsors can request significant designation either during the Investigational New Drug (IND) submission or as an amendment to an existing IND.
- : The FDA issues guidance documents that detail expectations for data submission and , which are critical for .
A comprehensive understanding of these regulations allows organizations to align their with FDA expectations, greatly improving their chances of receiving the innovative treatment designation. Significantly, from 2013 to 2023, the FDA authorized 157 original indications with exceptional designation, with 33% obtaining , highlighting the significance of following these guidelines.
Implement Compliance Strategies for Breakthrough Therapy Management
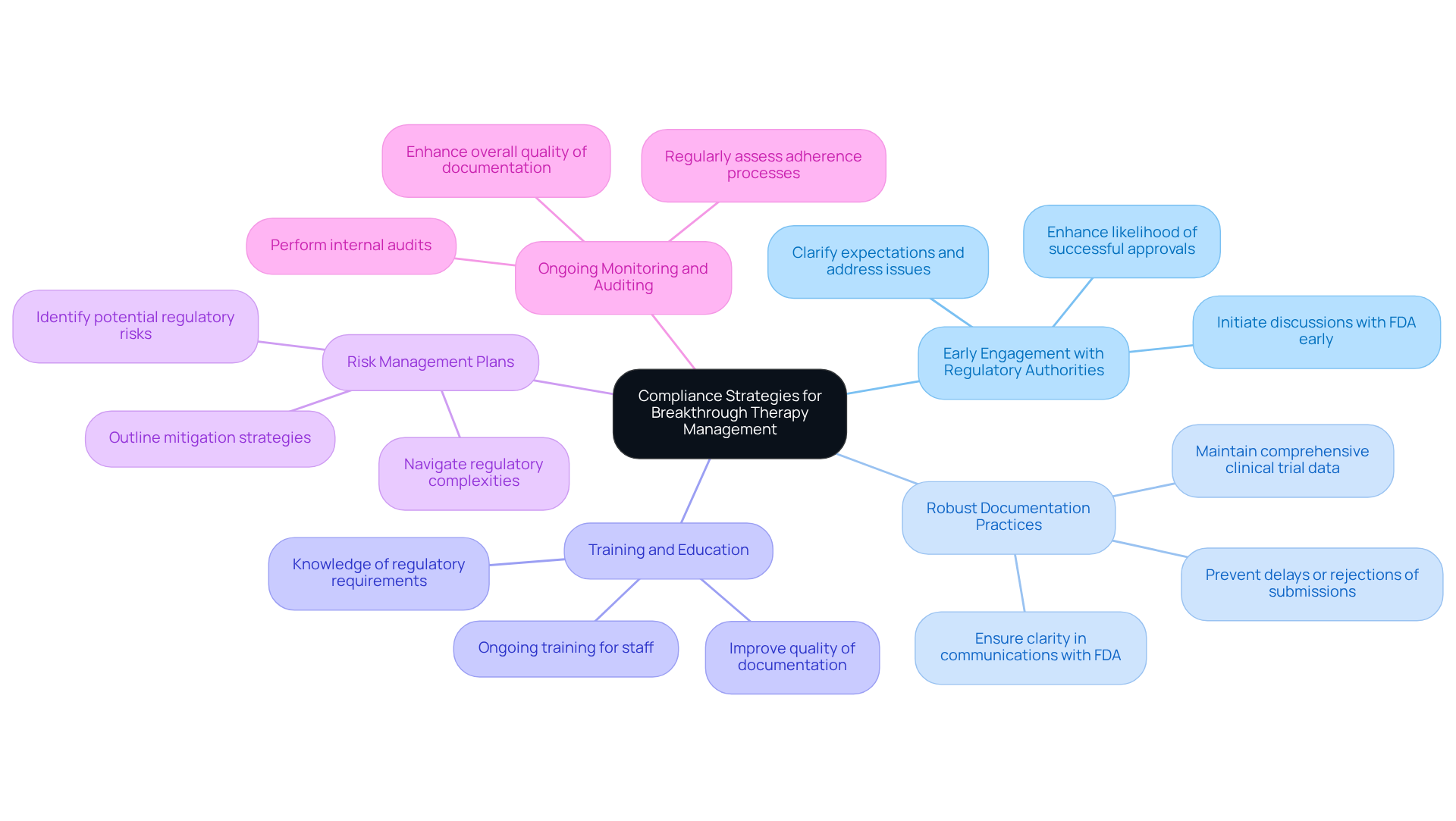
To effectively manage breakthrough therapies and ensure compliance, organizations must adopt the following strategies:
- : Initiate discussions with the FDA early in the development process to clarify expectations and proactively address potential issues. Engaging early can significantly enhance the likelihood of successful approvals; breakthrough-designated drugs have an approval rate of approximately 60% due to the guidance provided throughout development.
- : Maintain comprehensive documentation of all clinical trial data, communications with the FDA, and regulatory activities. This clarity is vital, as precise and thorough documentation is essential for and can prevent delays or rejections of submissions.
- Training and Education: Provide ongoing involved in the development and management of . Ensuring that team members are knowledgeable about and best practices is essential for upholding standards and improving the quality of documentation.
- : Create and execute risk management plans that identify potential regulatory risks and outline mitigation strategies. This proactive approach helps organizations navigate the complexities of regulatory requirements effectively.
- : Regularly assess adherence processes and perform internal audits to pinpoint areas for enhancement. Continuous monitoring ensures adherence to regulatory standards and enhances the overall quality of .
By implementing these approaches, organizations can strengthen their regulatory structures and increase their likelihood of effectively managing the intricacies of innovative treatment.
Address Challenges and Best Practices in Breakthrough Therapy Compliance
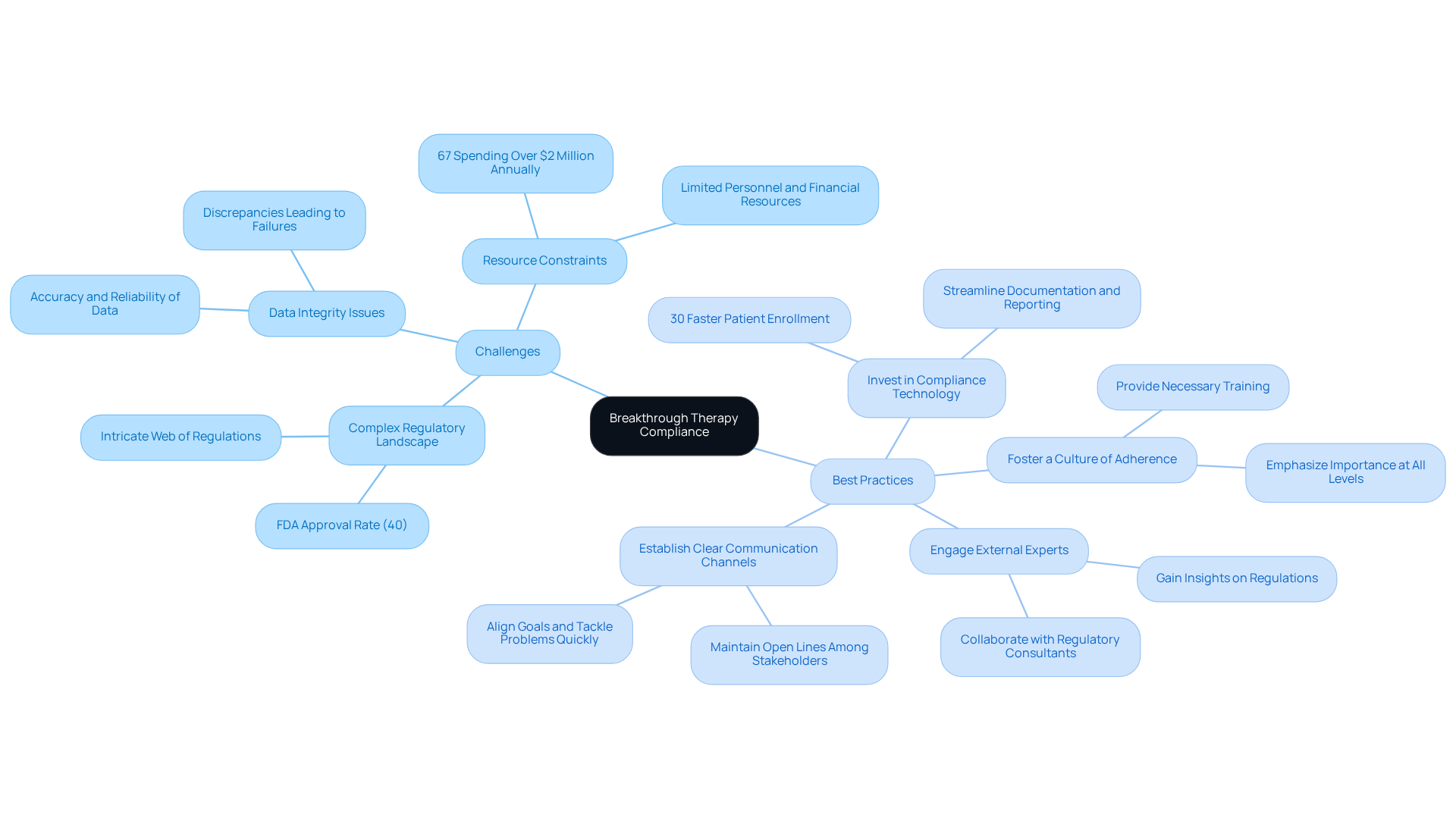
Organizations encounter significant challenges in ensuring compliance with breakthrough therapy regulations, including:
- : The intricate web of regulations can be overwhelming, particularly for smaller companies with limited resources. As of mid-2022, the FDA received 1,265 requests for Breakthrough Therapy Designation, with only around 40% granted, highlighting the competitive nature of .
- Issues: The accuracy and reliability of clinical trial data are crucial; discrepancies can lead to regulatory failures. Effective data governance strategies are essential, as they , ensuring that data integrity is maintained throughout the trial lifecycle.
- Resource Constraints: Limited personnel and financial resources can hinder the ability to uphold regulations and effectively manage breakthrough therapy. Approximately 67% of pharmaceutical companies report spending over $2 million annually on data privacy regulations, with projections indicating a 15-20% annual increase through 2025.
To address these challenges, organizations should adopt the following best practices:
- : Implement software solutions that streamline documentation, data management, and regulatory reporting. Clinical trials utilizing integrated, automated data management workflows complete patient enrollment 30% faster and achieve database lock in 45% less time than those using fragmented systems.
- : Cultivate a culture of adherence within the organization by emphasizing its importance at all levels and providing necessary training. This method not only improves adherence to regulations but also enables employees to prioritize following rules in their daily tasks.
- : Collaborate with or specialists to gain insights and guidance on navigating complex regulations. Their knowledge can assist organizations in avoiding typical challenges and simplifying the regulatory process.
- Establish Clear Communication Channels: Maintain open lines of communication among all stakeholders involved in the development process. Efficient communication is essential for aligning goals and quickly tackling any problems that emerge, ultimately resulting in enhanced adherence results.
By recognizing these challenges and implementing best practices, organizations can and enhance their chances of success in managing breakthrough therapy.
Conclusion
Understanding the intricacies of breakthrough therapy and its regulatory compliance is essential for stakeholders in the pharmaceutical industry. This designation not only accelerates the development and approval processes but also drives innovation, ultimately enhancing patient access to critical treatments. By aligning compliance strategies with regulatory expectations, organizations can significantly improve their chances of success in bringing life-saving therapies to market.
The article outlined key strategies for effective compliance management, including:
- Early engagement with regulatory authorities
- Robust documentation practices
- Ongoing training
- Implementation of risk management plans
Additionally, it addressed the challenges organizations face, such as:
- Navigating a complex regulatory landscape
- Maintaining data integrity
By adopting best practices, such as investing in compliance technology and fostering a culture of adherence, companies can streamline their operations and enhance their regulatory outcomes.
As the landscape of breakthrough therapies continues to evolve, it is crucial for organizations to remain proactive in their compliance efforts. Embracing these strategies not only facilitates adherence to regulations but also positions companies to better navigate the challenges that lie ahead. Ultimately, the commitment to robust compliance practices will play a pivotal role in the successful management of breakthrough therapies, ensuring that innovative treatments reach patients in need.
Frequently Asked Questions
What is breakthrough therapy in pharmaceuticals?
Breakthrough therapy is a designation awarded by the FDA to medications and biologics that demonstrate significant advancements over existing treatments for severe or life-threatening conditions.
Why is the breakthrough therapy designation important?
The designation is important because it accelerates both the development and review processes, allowing patients to access potentially life-saving treatments more quickly.
How does breakthrough therapy designation stimulate innovation?
It encourages pharmaceutical companies to invest in research for conditions with limited treatment options, fostering innovation in drug development.
What percentage of requests for breakthrough therapy designation have been granted as of mid-2024?
Approximately 38.7% of requests for breakthrough therapy designation have been granted as of mid-2024.
What services does AVS Life Sciences offer related to breakthrough therapy?
AVS Life Sciences offers regulatory compliance audits, quality management systems development, and training programs to assist clients in navigating compliance complexities associated with innovative treatments.
What is the FDA's commitment regarding requests for breakthrough therapy designation?
The FDA is committed to addressing requests for breakthrough therapy designation within 60 days, highlighting the program's significance in enhancing patient access to effective treatments.
How does breakthrough therapy designation impact late-stage clinical development time?
Breakthrough therapy designation has been shown to reduce late-stage clinical development time by 30%, significantly influencing pharmaceutical innovation.
Why is understanding the breakthrough therapy designation important for stakeholders?
Understanding the designation is essential for stakeholders to align their adherence strategies with regulatory expectations and ensure they meet the criteria for designation and subsequent approval.
