4 Steps to Choose the Right Compliance Consultant

Overview
The article delineates four crucial steps for selecting the appropriate compliance consultant:
- Identifying specific compliance needs
- Evaluating qualifications
- Conducting comprehensive assessments
- Grasping the consultant's approach to compliance challenges
Each step is bolstered by actionable insights, such as:
- The imperative for industry-specific experience
- The significance of tailored solutions
This framework equips organizations to adeptly navigate the regulatory landscape.
Introduction
Navigating the intricate world of regulatory compliance often resembles traversing a labyrinth, particularly for organizations within the pharmaceutical sector. The stakes are high, and the repercussions of missteps can be severe. Thus, selecting the right compliance consultant becomes a pivotal decision. This article presents a systematic approach to identifying and engaging a consultant who not only comprehends the complex regulatory landscape but also aligns with the unique needs of your organization.
As businesses endeavor to enhance their compliance frameworks, a critical question emerges: how can one ensure they are choosing a consultant capable of effectively bridging the gap between regulatory demands and organizational goals?
Identify Your Compliance Needs and Objectives
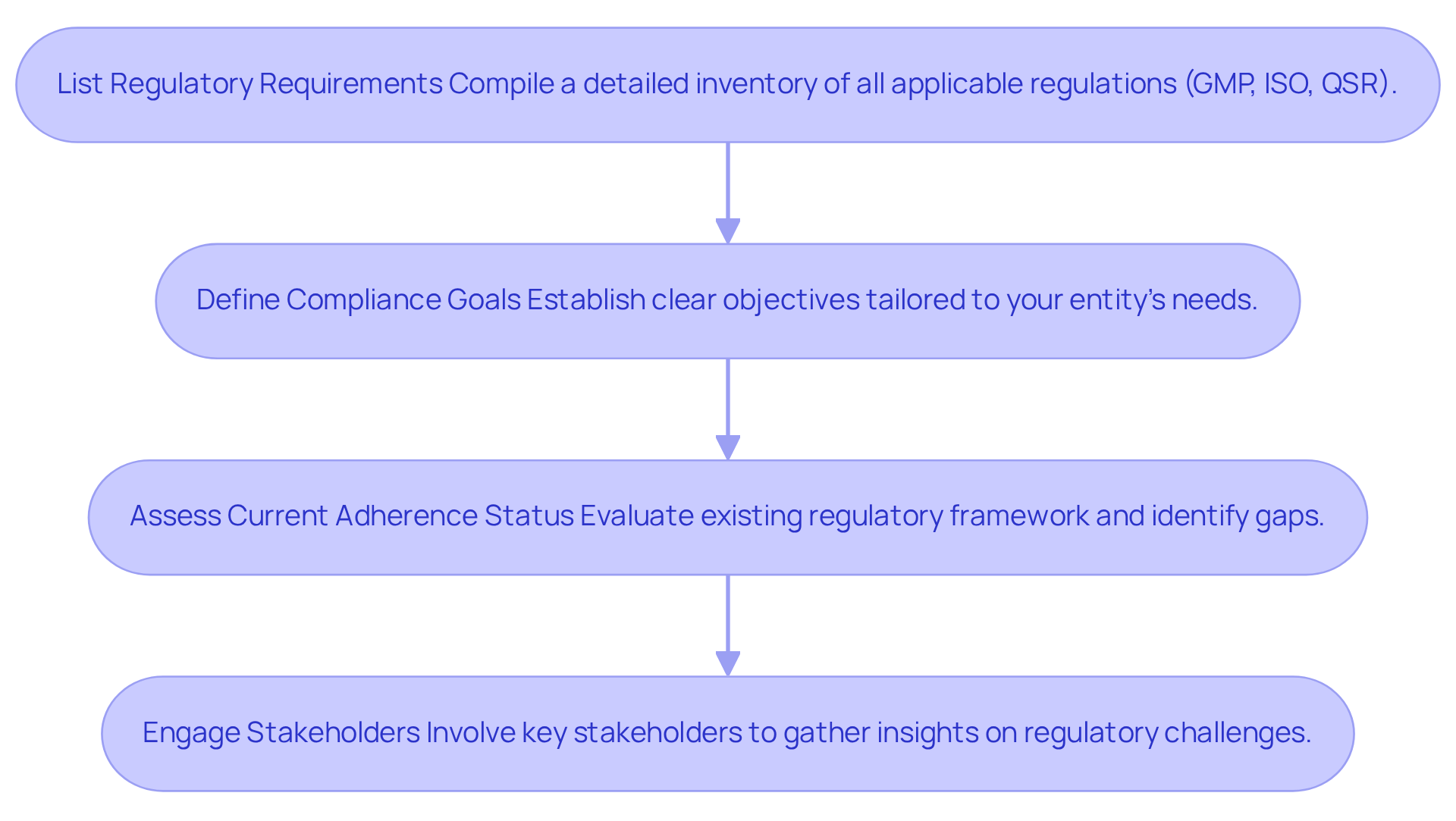
To successfully navigate in the pharmaceutical sector, begin with a comprehensive evaluation of your entity's specific legal environment. It is imperative to identify and understand key regulations, such as Good Manufacturing Practices (GMP), ISO standards, and Quality System Regulations (QSR). Follow these steps to ensure a thorough evaluation:
- List Regulatory Requirements: Compile a detailed inventory of all applicable regulations that your entity must adhere to. This includes GMP, ISO, and QSR standards, which are essential for compliance in the life sciences sector.
- Define Compliance Goals: Establish clear objectives tailored to your entity’s needs. These goals may encompass enhancing audit readiness, improving documentation practices, or ensuring comprehensive employee training programs.
- Assess Current Adherence Status: Conduct a thorough evaluation of your existing regulatory framework. Identify any gaps or areas needing enhancement, as 34% of entities currently outsource some regulatory functions, highlighting the necessity for robust internal evaluations.
- Engage Stakeholders: Involve key stakeholders from various departments to gather insights on regulatory challenges and objectives. This collaborative approach ensures that all perspectives are considered, fostering a culture of compliance throughout the organization.
By distinctly outlining and goals, you will be better positioned to select a compliance consultant who can effectively address these specific aspects. It is projected that by 2025, only 40% of adherence teams will invest in new technology for proactive oversight, underscoring the importance of staying ahead in a rapidly evolving governance environment.
Evaluate Qualifications and Industry Experience
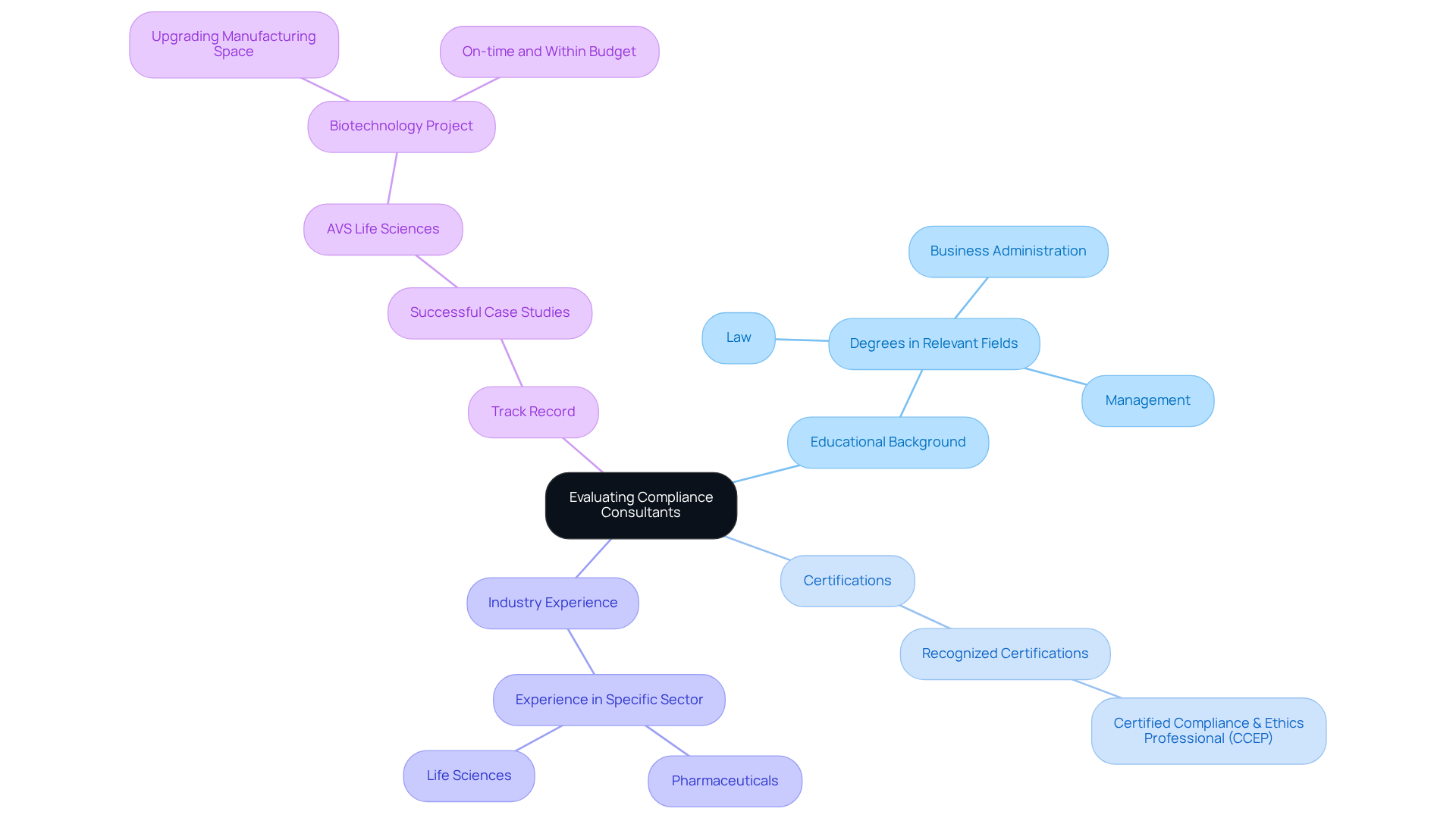
When evaluating potential compliance consultants, it is essential to consider several key criteria that can significantly impact your organization's success in regulatory compliance.
- Educational Background: Prioritize advisors with degrees in relevant fields such as law, business administration, or management. These qualifications provide a solid foundation for understanding complex legal frameworks.
- Certifications: Seek out recognized certifications, such as the Certified Compliance & Ethics Professional (CCEP). These credentials not only demonstrate expertise but also reflect a .
- Industry Experience: Assess their experience within your specific sector. For instance, a consultant with a background in pharmaceuticals will possess valuable insights into the unique challenges and requirements your company faces.
- Track Record: Request case studies or examples of previous projects that showcase their success in enhancing regulatory frameworks for organizations similar to yours. Notably, AVS Life Sciences has established itself as a trusted partner in the life sciences sector, recognized for its rapid value creation and thorough project execution. A significant case involved assisting a leading biotechnology company in upgrading its manufacturing space from a Biosafety Level 1 GMP facility to a Level 2 GMP facility. This project was completed on time and within budget, underscoring AVS's commitment to quality assurance and regulatory standards. Their approach included comprehensive gap analysis and meticulous documentation efforts to ensure full traceability, which were validated by the client’s quality assurance team. Such evidence of past performance instills confidence in their ability to deliver results.
By thoroughly evaluating these qualifications, you can ensure that the compliance consultant you choose possesses the essential expertise required to effectively navigate your organization through regulatory challenges.
Assess Potential Consultants Through Interviews and References
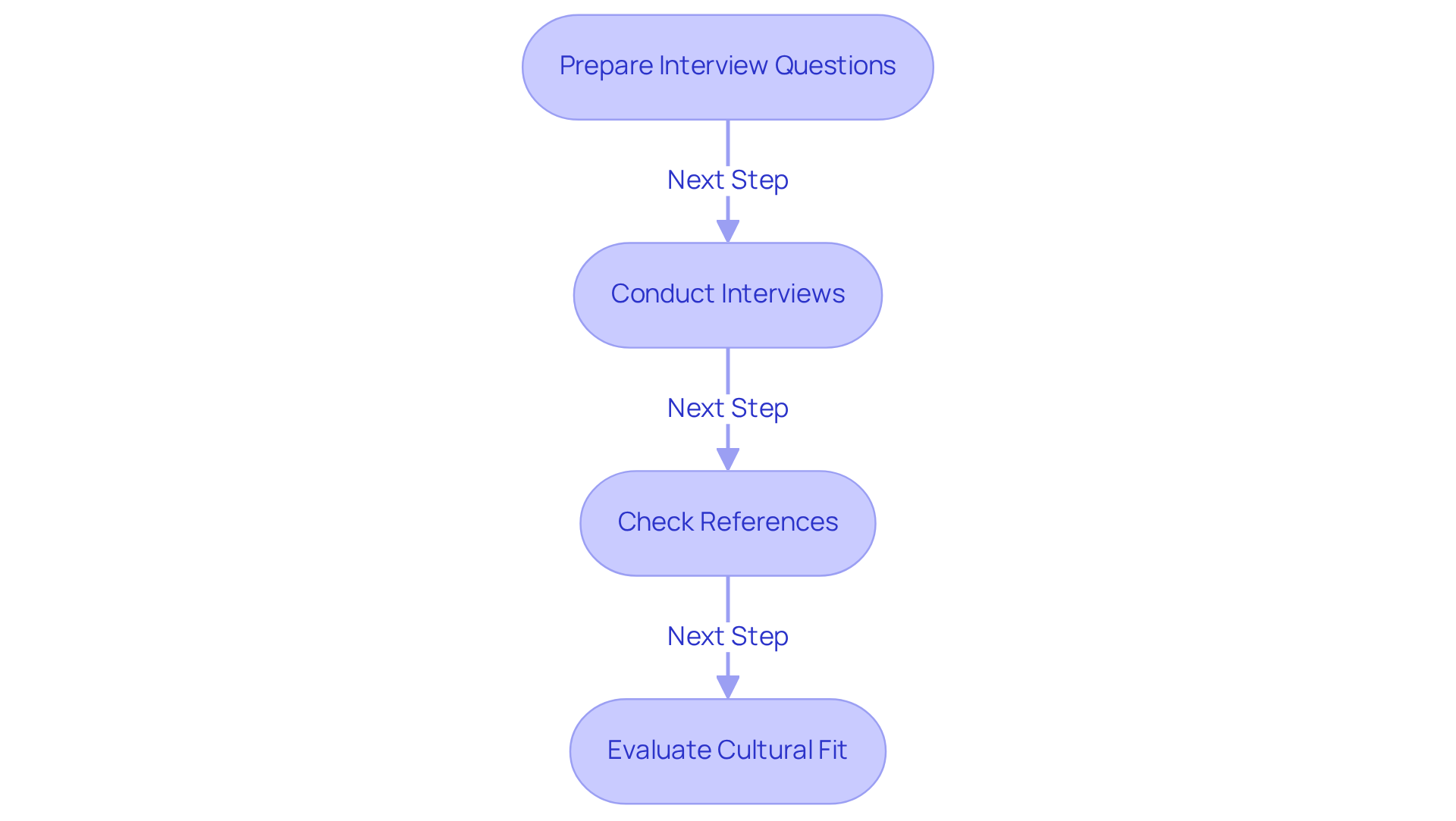
To effectively assess potential compliance consultants, follow these essential steps:
- Prepare Interview Questions: Develop a comprehensive list of inquiries that delve into the expert's methods regarding adherence, problem-solving techniques, and adaptability to regulatory changes. This preparation is crucial; a notable 92% of candidates assert that are vital for effective preparation.
- Conduct Interviews: Arrange interviews with shortlisted candidates to discuss your specific compliance needs. During these discussions, evaluate their responses and assess their understanding of industry standards, as 91% of employers favor candidates with relevant work experience.
- Check References: Engage with former clients to gather insights about their experiences with the advisor. Inquire about the advisor's effectiveness, communication skills, and ability to meet deadlines. Reference checks are essential, as they can reveal the advisor's history in managing regulatory challenges and ensuring client satisfaction.
- Evaluate Cultural Fit: Assess how well the individual's values align with your organization's culture. A strong cultural fit can significantly impact collaboration and project success; research indicates that 70% of job seekers lose interest in a role after reading negative reviews about a company.
By conducting thorough interviews and diligent reference checks, you can make a more informed choice about which advisor will best fulfill your requirements, ultimately enhancing your organization's ability to navigate complex legal landscapes. For customized consulting solutions in quality management and regulatory adherence, connect with AVS Life Sciences today!
Understand the Consultant's Approach to Compliance Challenges
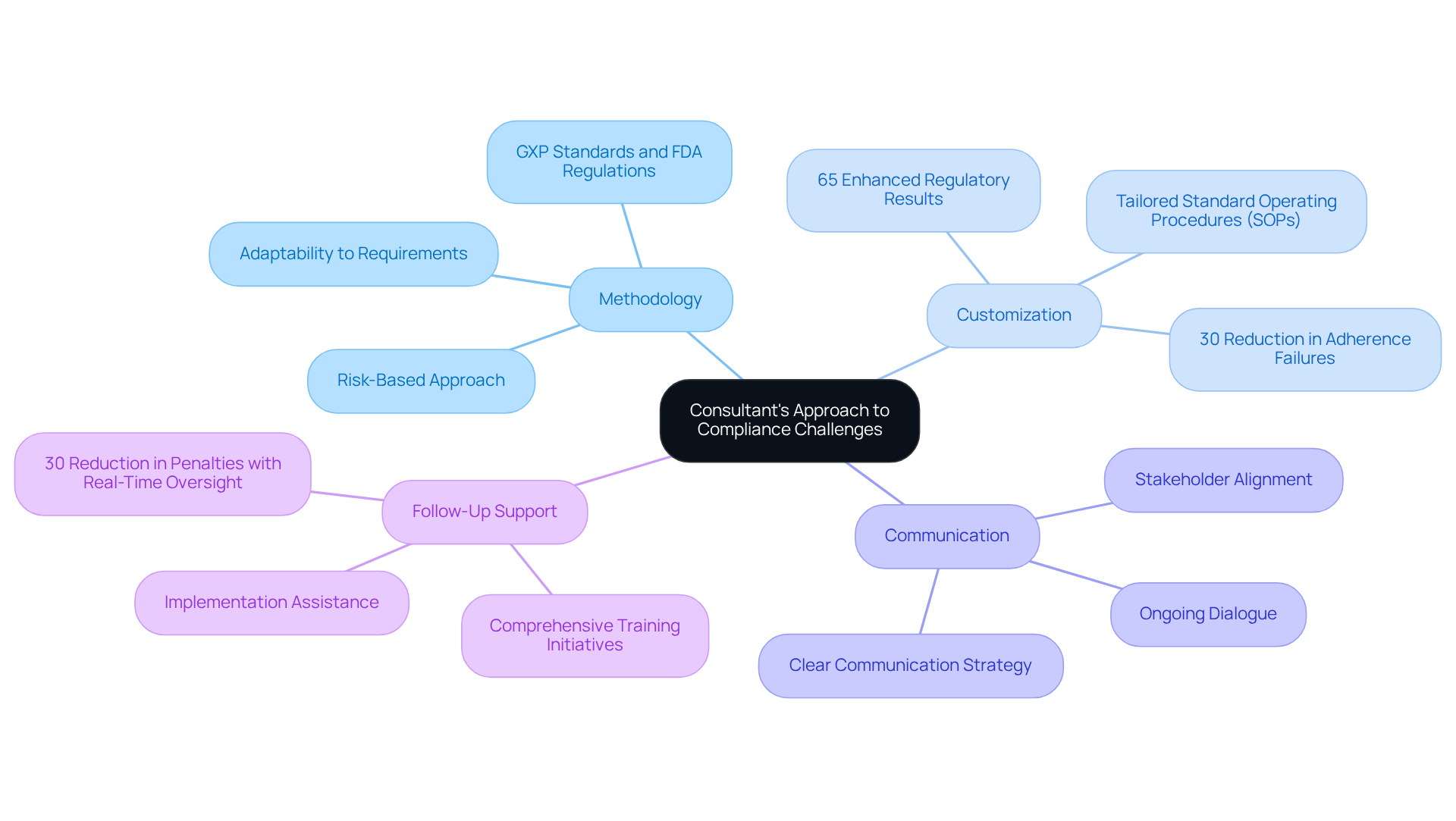
To gain insight into a consultant's approach, consider the following:
- Methodology: Inquire about their approach for assessing requirements and developing strategies. Do they utilize a risk-based method, or do they depend on verification checklists? Understanding their framework can reveal how adaptable they are to your specific requirements, especially when working with . AVS Life Sciences emphasizes a robust methodology that incorporates GXP standards and FDA regulations, ensuring that their strategies are aligned with industry best practices.
- Customization: Customization is essential in regulatory solutions. Data suggests that entities adopting customized adherence solutions can attain a 30% reduction in adherence failures. Furthermore, 65% of firms employing specialized advisors indicated enhanced regulatory results within the year. Inquire if customizes their approaches to suit your organization's distinct circumstances and difficulties, as this can greatly improve adherence effectiveness. AVS Life Sciences specializes in developing Standard Operating Procedures (SOPs) that are customized to meet the specific needs of biopharmaceuticals, medical devices, and nutraceuticals.
- Communication: Evaluate their communication strategy throughout the consulting process. Effective communication is essential for successful collaboration, ensuring that all stakeholders are aligned and informed. AVS Life Sciences prioritizes clear communication, facilitating ongoing dialogue to ensure that regulatory strategies are understood and effectively implemented.
- Follow-Up Support: Discuss the level of ongoing support they provide after the initial engagement. Will they help with the implementation and monitoring of adherence strategies? Ongoing assistance, encompassing comprehensive training initiatives, can be crucial for upholding adherence in a changing oversight landscape. As noted by the Compliance Inspections and Examinations Committee, entities with real-time oversight reduce penalties by 30% compared to those conducting periodic reviews. AVS Life Sciences provides thorough follow-up assistance, ensuring that clients are prepared to handle the intricacies of meeting legal requirements.
By grasping the strategy of the compliance consultant, especially their focus on personalization and adherence to GXP and FDA regulations, you can ensure that their methods align with your organization's regulatory goals and culture. Customized solutions not only address specific regulatory requirements but also foster a culture of compliance, leading to improved outcomes and reduced risks.
Conclusion
Selecting the right compliance consultant is a pivotal decision for any organization navigating the complex regulatory landscape. By systematically identifying compliance needs, evaluating qualifications, conducting thorough assessments, and understanding the consultant's approach, businesses can ensure they partner with an expert capable of addressing their unique challenges.
The importance of a comprehensive evaluation of an entity's regulatory requirements cannot be overstated. Engaging qualified professionals with relevant industry experience is essential, as is the critical role of effective communication and follow-up support. Each step outlined—from identifying objectives to assessing methodologies—serves to create a robust foundation for compliance success.
Ultimately, the right compliance consultant not only enhances regulatory adherence but also fosters a culture of compliance within the organization. By investing time and resources into this selection process, businesses can significantly mitigate risks and improve their operational integrity, ensuring they remain compliant in an ever-evolving regulatory environment. Embracing these strategies will empower organizations to thrive while maintaining the highest standards of compliance.
Frequently Asked Questions
What are the first steps to navigate the regulatory landscape in the pharmaceutical sector?
Begin with a comprehensive evaluation of your entity's specific legal environment, identifying and understanding key regulations such as Good Manufacturing Practices (GMP), ISO standards, and Quality System Regulations (QSR).
How can I ensure a thorough evaluation of compliance needs?
To ensure a thorough evaluation, compile a detailed inventory of all applicable regulations, establish clear compliance goals, assess your current adherence status, and engage stakeholders from various departments.
What should be included in the list of regulatory requirements?
The list of regulatory requirements should include all applicable regulations that your entity must adhere to, specifically GMP, ISO, and QSR standards essential for compliance in the life sciences sector.
How do I define compliance goals for my entity?
Compliance goals should be tailored to your entity’s needs and may include objectives such as enhancing audit readiness, improving documentation practices, or ensuring comprehensive employee training programs.
Why is it important to assess the current adherence status?
Assessing the current adherence status helps identify gaps or areas needing enhancement in your regulatory framework, as 34% of entities currently outsource some regulatory functions, highlighting the need for robust internal evaluations.
What is the benefit of engaging stakeholders in the compliance process?
Engaging stakeholders from various departments allows for gathering insights on regulatory challenges and objectives, fostering a culture of compliance throughout the organization.
How can I select a compliance consultant effectively?
By distinctly outlining your regulatory requirements and goals, you will be better positioned to select a compliance consultant who can effectively address these specific aspects.
What is projected regarding adherence teams and technology investment by 2025?
It is projected that by 2025, only 40% of adherence teams will invest in new technology for proactive oversight, emphasizing the importance of staying ahead in a rapidly evolving governance environment.
