4 Key Practices for Oligonucleotide Therapeutics Compliance

Overview
This article delves into the essential practices necessary for ensuring compliance in the development and application of oligonucleotide therapeutics. It underscores the critical importance of adhering to regulatory guidelines, implementing robust quality management systems, and enhancing training and documentation practices. These elements are vital for effectively navigating the complex landscape of oligonucleotide therapies.
By focusing on these practices, organizations can not only meet compliance challenges but also foster a culture of excellence in therapeutic development. Engaging with these strategies will empower stakeholders to proactively address compliance issues and drive successful outcomes in their projects.
Introduction
Oligonucleotide therapeutics are revolutionizing modern medicine, providing innovative solutions to complex genetic disorders through targeted gene expression modulation. As the market for these therapies expands rapidly, understanding the intricacies of compliance becomes paramount for developers and manufacturers alike.
The path to regulatory adherence, however, is fraught with challenges. Organizations face the daunting task of navigating evolving guidelines while implementing robust quality management systems.
How can they effectively ensure compliance while maximizing the therapeutic potential of these groundbreaking treatments? This question underscores the need for strategic engagement with compliance solutions that not only meet regulatory standards but also enhance the efficacy of these revolutionary therapies.
Understand Oligonucleotide Therapeutics: Definitions and Types
represent short, synthetic strands of nucleic acids meticulously designed to influence , thereby playing a crucial role in modern medicine. These treatments can be classified into several key types:
- Antisense Oligonucleotides (ASOs): These oligonucleotides are specifically engineered to bind to targeted mRNA molecules, effectively preventing their translation into proteins. This mechanism proves particularly valuable in treating by silencing detrimental genes. The market for oligonucleotide therapeutics, specifically , is poised for significant growth, projected to expand at a 15.3% CAGR from 2025 to 2034, starting with a market size valued at USD 2.2 Billion in 2024 and anticipated to reach USD 9.1 Billion by 2034.
- Small Interfering RNAs (siRNAs): siRNAs facilitate RNA interference, a natural cellular process that culminates in the degradation of target mRNA. This method has shown successful applications in clinical trials, particularly for conditions such as cancer and viral infections, where downregulating specific genes can yield substantial health benefits.
- Aptamers: Aptamers are versatile oligonucleotides that bind to specific proteins or other molecules, inhibiting their function. Their adaptability allows for use in various treatment contexts, including targeted drug delivery and diagnostics.
Understanding these definitions and categories is vital for navigating the . However, the market is not without its challenges, including , as well as . The increasing prevalence of genetic disorders and the demand for innovative treatments further underscore the in contemporary medicine. Currently, the number of approved oligonucleotide therapeutics continues to rise, reflecting the growing recognition of their potential in addressing a wide array of illnesses. This expansion is supported by advancements in technology and ongoing research, which are essential for ensuring and achieving .

Navigate Regulatory Frameworks for Oligonucleotide Development
Navigating the compliance environment for oligonucleotide treatments presents a complex challenge that demands meticulous attention to detail. Key considerations include:
- : Recent FDA guidance documents offer essential insights into the development and evaluation of oligonucleotide therapeutics, addressing both non-clinical and clinical aspects crucial for successful product advancement. Notably, there have been 22 FDA- and/or EMA-approved to date, reflecting the evolving oversight landscape.
- : The European Medicines Agency has established comprehensive guidelines that must be adhered to for products targeting the European market, ensuring that all are satisfied.
- : Adherence to is vital for ensuring . Companies are required to maintain meticulous records and implement stringent quality control measures throughout the .
Furthermore, challenges in developing oligonucleotide therapeutics at the Chemistry, Manufacturing, and Controls (CMC), nonclinical, and clinical levels must be addressed to effectively navigate this intricate landscape. Remaining informed about these is imperative for achieving successful .

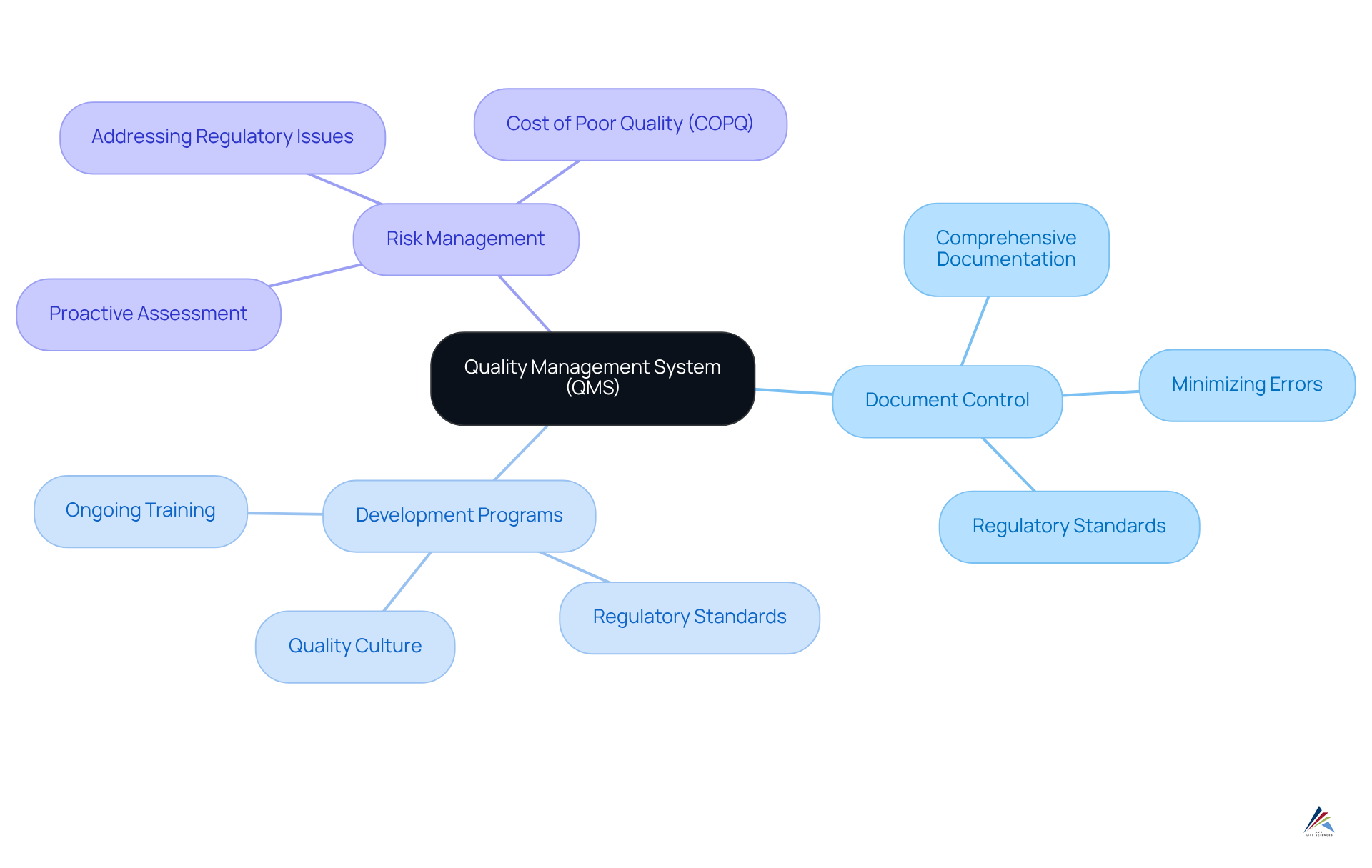
Implement Quality Management Systems for Compliance Assurance
A robust is essential for ensuring adherence in oligonucleotide therapeutics. Document Control is a critical component, as comprehensive documentation of all processes—spanning development, manufacturing, and testing—is vital. Efficient assist organizations in preserving clarity and consistency, minimizing the likelihood of errors and ensuring adherence to regulatory standards. Companies that effectively implement quality management practices experience a 26% increase in profitability, highlighting the .
Development Programs are equally crucial; ongoing instruction for personnel on fosters a culture of quality. AVS Life Sciences offers that ensure all team members are well-versed in the latest protocols and regulations, which is essential for maintaining high standards in product development. This ongoing education is vital, as organizations with mature report a .
involves implementing that enable organizations to identify and address potential regulatory issues before they escalate. By addressing risks early, companies can improve their adherence frameworks and significantly lessen the chances of findings during audits. Notably, the (COPQ) can range from 5% to 30% of sales revenue, underscoring the financial implications of neglecting a robust QMS.
Incorporating these components into a QMS not only enhances compliance but also enables organizations to react effectively to the changing oversight environment in the life sciences field. Companies that prioritize these practices are more likely to achieve higher customer satisfaction and operational efficiency, ultimately leading to improved market performance. However, it is essential to be aware of potential pitfalls during QMS implementation, such as resistance to change and insufficient preparation, to ensure a successful transition.
Enhance Training and Documentation Practices for Regulatory Adherence
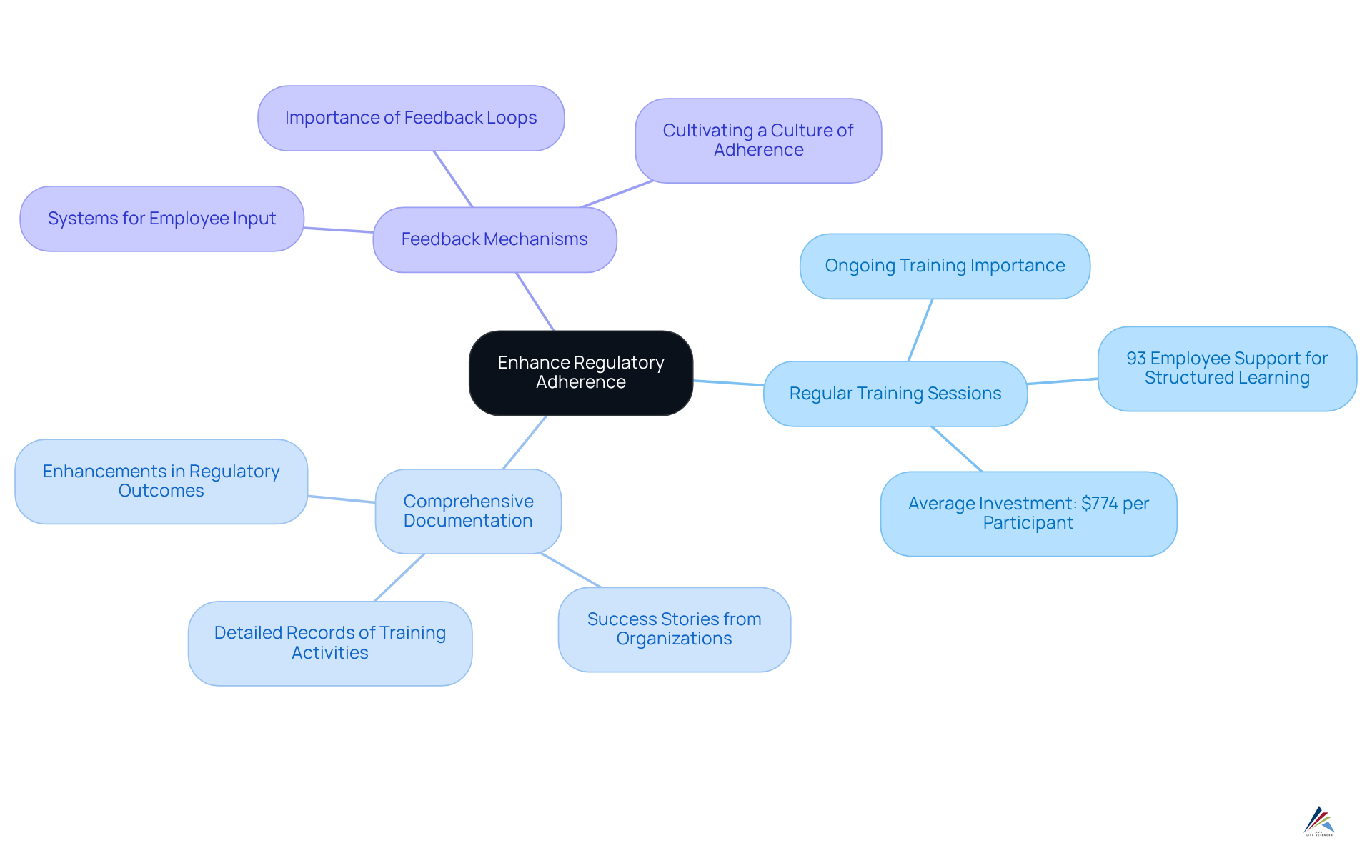
To enhance , organizations must prioritize robust training and documentation practices:
- Regular Training Sessions: Ongoing training for all employees involved in the development and manufacturing of oligonucleotide therapeutics is essential. This ensures that staff remain informed about the latest regulatory requirements and best practices. Statistics reveal that 93% of employees believe positively influence engagement, underscoring the necessity for structured learning initiatives. In 2024, organizations in the United States allocated an average of $774 per participant for employee education and development, highlighting the importance of investing in development programs.
- : Maintaining is crucial. This includes documentation of attendance, materials covered, and assessments. Thorough documentation not only aids in meeting regulatory requirements but also serves as a valuable resource for audits and inspections. Success stories from organizations that have adopted thorough documentation practices demonstrate considerable enhancements in regulatory outcomes. For instance, companies that view have reported enhanced compliance and lower turnover rates.
- Feedback Mechanisms: Implementing systems to gather input from employees about the effectiveness of the development programs is vital. This enables organizations to identify areas for enhancement and modify development programs to more effectively address the requirements of their workforce. Regulatory officers stress that thorough documentation and feedback loops are essential to cultivating a culture of adherence. As one regulatory officer noted, "[Comprehensive documentation](http://avslifesciences.com/blog-post/10-essential-insights-for-pharma-regulatory-compliance-success) and regular feedback are essential for ensuring that our remain relevant and effective."
By prioritizing these practices, organizations can cultivate a culture of compliance, ensuring that all team members are well-equipped to meet regulatory expectations and effectively contribute to the success of oligonucleotide therapeutics.
Conclusion
Oligonucleotide therapeutics signify a transformative leap in modern medicine, providing targeted interventions for a spectrum of genetic disorders and diseases. The successful development and implementation of these therapies depend on a thorough understanding of their classifications, coupled with strict adherence to regulatory frameworks and quality management practices. By adopting these critical practices, organizations can ensure compliance while simultaneously boosting the overall efficacy of their therapeutic offerings.
The article delineated four fundamental practices essential for compliance in oligonucleotide therapeutics:
- Grasping the various types of oligonucleotides
- Navigating intricate regulatory frameworks
- Instituting robust quality management systems
- Refining training and documentation practices
Each of these components is pivotal in cultivating a culture of compliance, which is indispensable for the successful development and market approval of these pioneering therapies. As the market for oligonucleotide therapeutics continues to grow, the necessity for meticulous attention to these practices becomes increasingly apparent.
In conclusion, the importance of adhering to best practices in oligonucleotide therapeutics cannot be overstated. Organizations are urged to prioritize continuous education, rigorous documentation, and proactive risk management strategies to adeptly navigate the evolving landscape of regulatory compliance. By doing so, they not only protect their operations but also contribute to the progression of life-saving therapies that hold the potential to revolutionize patient care. Investing in compliance today is a strategic investment in the future of medicine.
Frequently Asked Questions
What are oligonucleotide therapeutics?
Oligonucleotide therapeutics are short, synthetic strands of nucleic acids designed to influence gene expression, playing a crucial role in modern medicine.
What are the main types of oligonucleotide therapeutics?
The main types include Antisense Oligonucleotides (ASOs), Small Interfering RNAs (siRNAs), and Aptamers.
How do Antisense Oligonucleotides (ASOs) work?
ASOs are engineered to bind to targeted mRNA molecules, preventing their translation into proteins, which is valuable for treating genetic disorders by silencing harmful genes.
What is the projected market growth for Antisense Oligonucleotide Therapeutics?
The market is projected to grow at a 15.3% CAGR from 2025 to 2034, starting from a value of USD 2.2 Billion in 2024 and expected to reach USD 9.1 Billion by 2034.
What role do Small Interfering RNAs (siRNAs) play in medicine?
siRNAs facilitate RNA interference, leading to the degradation of target mRNA, and have been successfully used in clinical trials for conditions like cancer and viral infections.
What are Aptamers and their applications?
Aptamers are versatile oligonucleotides that bind to specific proteins or molecules to inhibit their function, used in targeted drug delivery and diagnostics.
What challenges does the oligonucleotide therapeutics market face?
The market faces challenges such as high development and manufacturing costs, as well as regulatory hurdles.
Why are oligonucleotide therapeutics important in contemporary medicine?
They are important due to the increasing prevalence of genetic disorders and the demand for innovative treatments, with a rising number of approved therapeutics reflecting their potential in addressing various illnesses.
