What Are Pharmaceutical Compliance Consulting Services? A Comprehensive Overview

Introduction
In the intricate realm of pharmaceuticals, where safety and efficacy stand as imperatives, compliance transcends mere regulatory obligation—it is the bedrock of public trust. As organizations navigate a perpetually shifting landscape of rigorous regulations, the importance of compliance consulting has reached unprecedented heights. These specialized advisory services provide indispensable support, steering companies through the complexities of:
- Good Manufacturing Practices (GMP)
- Quality System Regulations (QSR)
- International compliance standards
With startling statistics indicating that nearly all organizations have encountered compliance breaches in recent years, the necessity for robust frameworks becomes glaringly apparent. This article explores the critical role of pharmaceutical compliance consulting, examining its advantages, challenges, and the transformative effects it can exert on operational efficiency and product quality within the life sciences sector.
Understanding Pharmaceutical Compliance Consulting Services
encompass a diverse range of specialized advisory roles designed to ensure that pharmaceutical firms adhere to stringent legal standards and best practices. These services provide expert guidance on , , and International Organization for Standardization (ISO) standards. AVS Life Sciences delivers extensive , focusing on quality adherence and tailored validation solutions for the pharmaceutical and biotechnology sectors.
Regulatory advisors collaborate closely with organizations to formulate and implement strategies that not only satisfy legal obligations but also enhance operational efficiency and product quality.
The significance of is underscored by alarming statistics indicating that involving third parties in the past two years. This statistic highlights the critical need for robust regulatory frameworks, as effective consulting can mitigate such risks. As the pharmaceutical regulatory landscape grows increasingly complex, compliance consulting services are essential in safeguarding public health and ensuring that companies can successfully introduce safe and effective products to the market.
Current industry trends reveal a heightened focus on proactive adherence strategies, with many organizations acknowledging the necessity of integrating into their overall business operations. This shift is evident in the rising demand for advisory consultants who can provide customized solutions addressing specific legal challenges. Notably, a recent report from Thomson Reuters indicates that 61% of participants expect the costs associated with senior oversight officers to increase, further emphasizing the evolving nature of roles within the industry.
Key functions of [pharmaceutical compliance consulting services](https://drata.com/blog/compliance-statistics) encompass conducting thorough audits, developing comprehensive oversight programs, and offering training to ensure staff are well-versed in legal requirements. AVS excels in these areas, providing expert solutions in GMP adherence, validation, and engineering, which are crucial for maintaining product integrity and quality. Additionally, AVS Health prioritizes essential elements such as , Standard Operating Procedures (SOPs) Development, and CFR Part 11 Compliance to ensure comprehensive regulatory adherence.
A transformative case study illustrates AVS Health's successful enhancement of a biotechnology GMP facility, where the company assisted a leading San Francisco-based biotechnology firm in transitioning from a Biosafety Level 1 GMP facility to a Level 2 GMP facility. This project was completed on time and within budget, with documentation efforts deemed satisfactory by the client’s quality assurance team. Such collaborations empower clients to focus on their core mission—developing medications to improve patient quality of life—while AVS guarantees adherence and quality assurance throughout the process.
This case study underscores AVS Life's commitment to delivering customized compliance solutions that directly address their clients' needs.
Expert insights emphasize that the effectiveness of pharmaceutical compliance consulting services can significantly impact pharmaceutical companies, leading to improved operational outcomes and reduced risk of penalties. Clients of AVS Life Sciences value the thoroughness and professionalism of its consultants, which contributes to timely project delivery and zero findings during audits. This dedication to quality not only enhances client satisfaction but also positions firms as leaders in the competitive life sciences industry.
Moreover, prevalent warning letter citations in compliance matters, as analyzed by specialists at Lachman Consultants, highlight the challenges faced by pharmaceutical companies. Regulatory advisors play a crucial role in guiding organizations through these challenges and helping them avoid potential pitfalls.
In summary, the role of regulatory consultants in the pharmaceutical sector is indispensable, as they provide compliance consulting services that navigate firms through the intricate regulatory environment, ensuring that companies not only comply with existing standards but also adapt to emerging trends and challenges.

The Importance of Compliance in the Pharmaceutical Industry
Compliance in the pharmaceutical industry is essential due to the inherent risks associated with drug development and manufacturing. Regulatory agencies, including the FDA and EMA, enforce rigorous guidelines designed to ensure that products are safe, effective, and produced to the highest quality standards. The repercussions of non-compliance can be severe, encompassing , legal repercussions, and the potential loss of market access.
For instance, recent FDA enforcement actions have underscored the repercussions encountered by companies that neglect to follow established regulations, leading to millions of dollars in fines and considerable reputational harm. Furthermore, failures to meet standards can have severe effects on patient safety, potentially resulting in negative health outcomes and a substantial decline in public confidence in pharmaceutical products. Statistics indicate that businesses can by appointing a dedicated compliance leader, highlighting the . Engaging is essential for companies aiming to mitigate these risks and uphold their commitment to quality and safety.
A transformative case study involving AVS Health effectively illustrates this point. AVS Healthcare assisted a prominent biotechnology firm in from a Biosafety Level 1 GMP facility to a Level 2 GMP facility, completing the project on time and within budget. This upgrade not only improved the client's capability to produce essential medications but also ensured that their quality assurance procedures were robust and adhered to regulatory standards.
The case study emphasizes how AVS's thorough quality management and allowed the client to concentrate on their primary mission: creating medicines that enhance patient quality of life. During the upgrade process, AVS Biological discovered anomalies in test results due to barcode scanner cameras being installed upside down, which had previously produced misleading results. This oversight was addressed, and important lessons learned were captured, leading to a reevaluation of the client's business processes to enhance reliability in test results.
By tackling and ensuring complete traceability, AVS Life Sciences assisted the client in enhancing their , ultimately resulting in better data integrity and reduced noncompliance rates.
Industry experts stress that the significance of adherence in drug development cannot be overstated. , including the potential for product recalls and increased scrutiny from regulatory bodies. Richard Stevenson, a specialist in cybersecurity risk management, observes that developing robust regulatory programs is crucial for fulfilling security requirements and effectively managing risks.
As the landscape of pharmaceutical regulations continues to evolve, the need for strong adherence strategies becomes increasingly critical. By prioritizing adherence to standards, pharmaceutical firms can not only evade the hazards of oversight failures but also cultivate a culture of quality that benefits both their operations and the patients they assist.
AVS is positioned as a leader in due to its commitment to excellence and client satisfaction, making it a trusted partner for companies aiming to improve their adherence efforts.

Key Services Offered by Pharmaceutical Compliance Consultants
offer a comprehensive suite of solutions meticulously designed to meet the specific needs of clients navigating a complex oversight environment. Key offerings include:
- : At AVS, consultants develop thorough strategies that align with current regulations and anticipate future changes, ensuring clients remain compliant and competitive. This proactive approach is crucial; Acemoglu and Linn (2003) estimate that each . This underscores the significance of effective oversight strategies.
- : AVS consultants are instrumental in preparing companies for compliance audits and inspections. By meticulously organizing all documentation and processes, they significantly enhance the likelihood of successful audit outcomes. Businesses that engage in comprehensive audit preparation often report success rates that exceed industry norms, highlighting the value of professional guidance.
- Training and Education: Focused training initiatives for employees are essential for fostering a culture of compliance within organizations. AVS Health Solutions offers programs that equip employees with the necessary knowledge to navigate adherence requirements and implement best practices efficiently.
- : Consultants assist organizations in establishing and maintaining robust quality management systems that meet industry standards, including GXP and FDA guidelines. This has become increasingly vital as the pharmaceutical compliance sector evolves, particularly with the anticipated growth in Latin America due to the influx of biopharmaceutical firms. For instance, AVS Life Solutions provides alongside validation and commissioning, quality adherence consulting, and submissions guidance to support these initiatives.
- Risk Evaluation and Mitigation: Conducting thorough assessments to identify potential compliance risks is a core service. AVS consultants develop tailored strategies to mitigate these risks, ensuring organizations can operate smoothly and avoid costly legal challenges.
These services are critical not only for maintaining standards but also for enhancing overall operational efficiency. As the landscape of pharmaceutical regulations becomes increasingly intricate, the expertise of regulatory consultants is invaluable in helping organizations navigate these challenges successfully. Recent case studies, such as AVS's successful upgrade from Biosafety Level 1 to Level 2 GMP facility for lentivirus production, showcase ongoing efforts to expand service capabilities and geographical reach, further emphasizing the importance of strategic partnerships in Regulatory Affairs.
With the anticipated increase in demand for legal representation services due to complex healthcare regulations, the role of regulatory consultants will continue to grow in significance. AVS Healthcare, recognized for its extensive industry expertise and an impressive 80% repeat business rate, positions itself as a trusted partner in this evolving landscape. Ready to enhance your compliance strategies?
today to initiate our expert consulting services tailored for the life sciences industry.
Navigating the Regulatory Landscape in Pharmaceuticals
The governing framework in the drug industry presents significant challenges for firms striving to uphold . With the Food and Drug Administration (FDA) overseeing in the United States and the European Medicines Agency (EMA) fulfilling a similar role in Europe, companies must navigate a complex and ever-evolving landscape. As of April 2025, both agencies have enacted updates to their oversight frameworks, demonstrating a commitment to enhancing drug safety and efficacy.
Compliance with local regulations is crucial, yet these can vary considerably across different jurisdictions. This complexity underscores the critical need for advisors specializing in within the healthcare industry. These specialists provide essential assistance by keeping organizations informed about compliance changes, preparing them for inspections, and ensuring that their products meet all necessary requirements. For instance, U.S. drug companies allocate over 21% of their revenues to research and development, with R&D spending peaking at $102 billion in 2021.
Despite a minor decline in 2023, the sector's dedication to innovation remains strong, necessitating effective oversight approaches to successfully launch new medications into the market. Moreover, specialty product expenditure surged by 11.7% in 2022, highlighting the industry's growth and the importance of adherence in this expanding market.
As the pharmaceutical industry progresses in its digital transformation strategies in 2024, there is a growing focus on the digitization and automation of compliance and quality control processes, which are supported by . This shift aims to improve accuracy and effectiveness, further emphasizing the role of compliance consultants in facilitating these transitions. As Sarah Lee observed, "Pharmaceutical companies using predictive analytics for compliance risk management have reported a 40% reduction in adherence violations and a 25% decrease in audit findings."
By offering , consultants enable companies to concentrate on their primary business goals while significantly reducing the risk of legal violations. A notable example is AVS Health's collaboration with a prominent biotechnology firm in San Francisco, where they successfully upgraded the client's production area from a Biosafety Level 1 GMP facility to a Level 2 GMP facility. This project was completed on time and within budget, showcasing AVS's commitment to and adherence to regulations. Their meticulous documentation efforts ensured full traceability, validated by the client's quality assurance team.
During the upgrade, AVS Health employed methodologies such as gap analysis and risk evaluation to identify potential early in the process. Challenges included addressing anomalies in test results due to improperly installed barcode scanners, which were resolved through enhanced training and process adjustments. This partnership allowed the client to focus on developing innovative medicines, ultimately improving patient outcomes.
AVS Life Sciences stands as a leader in the life sciences consulting market by offering . Their dedication to excellence and client satisfaction makes them a valuable partner for organizations navigating these challenges.
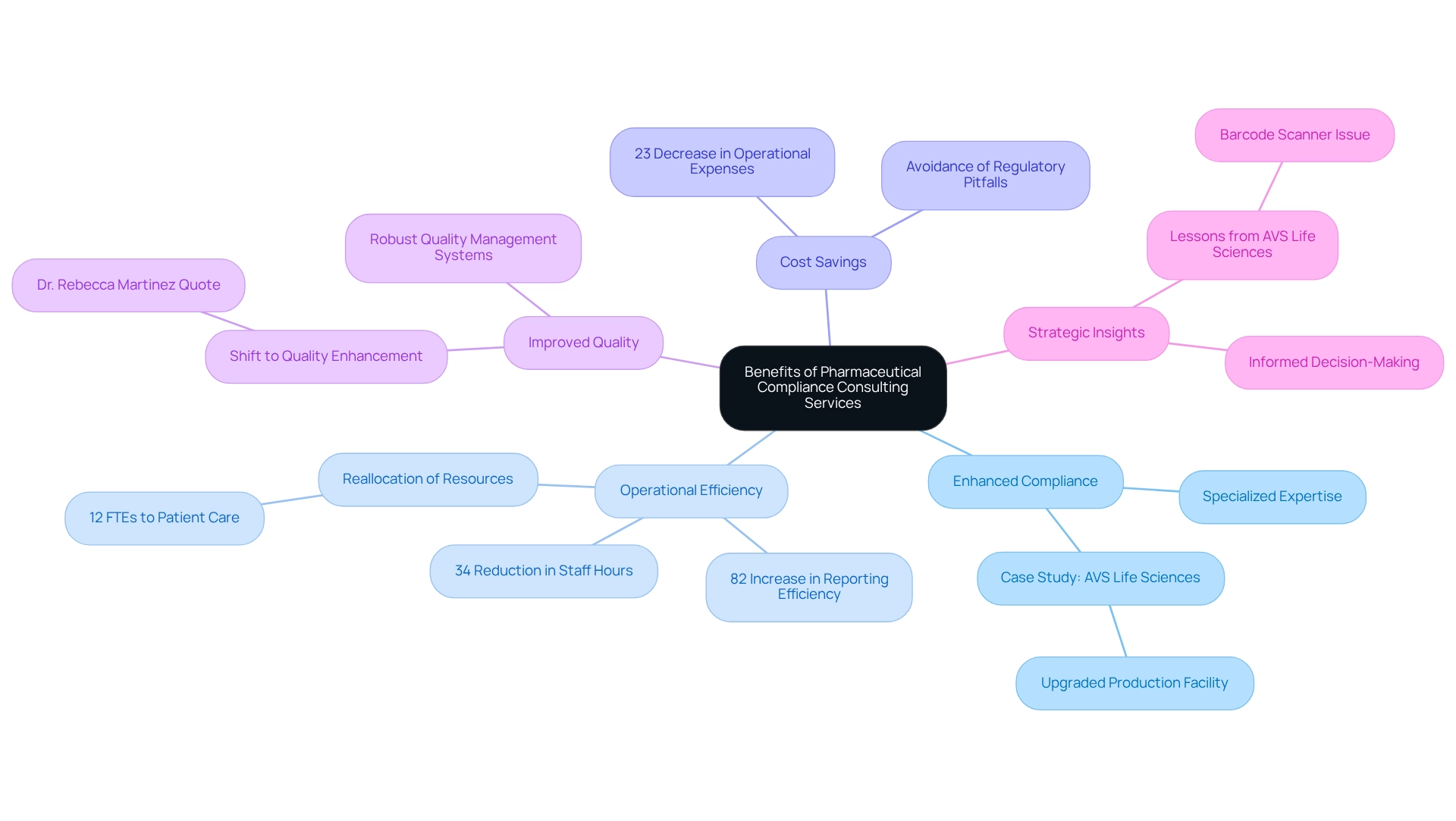
Benefits of Engaging Pharmaceutical Compliance Consulting Services
Engaging presents a multitude of advantages that can significantly impact an organization’s operational landscape:
- Enhanced Compliance: Compliance consultants offer specialized expertise to navigate the intricate web of regulations, thereby . This is vital in an industry characterized by intense oversight, where the repercussions of non-compliance can be severe. For instance, AVS Life Sciences effectively supported a prominent biotechnology firm in upgrading their production area from a Biosafety Level 1 GMP facility to a Level 2 GMP facility, underscoring their commitment to quality assurance and .
- Operational Efficiency: By streamlining processes and implementing industry best practices, advisory consultants can drive substantial improvements in operational efficiency. A study revealed that organizations utilizing , alongside a 34% reduction in staff hours dedicated to reporting tasks. This allows resources to be , such as patient care. A medium-sized hospital system reported reallocating 12 full-time equivalent positions from manual reporting to patient care initiatives after implementing these solutions.
- Cost Savings: The financial consequences of regulatory issues can be staggering. Involving regulatory consultants and leveraging aids in avoiding , legal expenses, and product recalls. In fact, across various functions, including . 'AVS Consulting' services are tailored to assist clients in realizing substantial cost reductions, as demonstrated by their successful project delivery within budget and on time for their biotechnology client.
- Improved Quality: Consultants play a pivotal role in establishing robust that and ensure safety. This commitment to quality is increasingly recognized as a strategic advantage in the marketplace. As Dr. Rebecca Martinez, a former CMS Innovation Center advisor, noted, "We’re witnessing a fundamental shift from adherence as documentation to adherence as demonstrable quality enhancement—analytics is the bridge making this transition possible." AVS Health exemplifies this change through their comprehensive quality management solutions.
- Strategic Insights: With their extensive industry knowledge, regulatory advisors provide invaluable insights that inform and enhance market positioning. This perspective is supported by analytics, serving as a bridge to achieving demonstrable quality enhancements. The lessons learned from AVS Life Sciences' collaboration with their client led to a reevaluation of business processes, ultimately improving the reliability of test results and fostering a culture of accountability within the organization. Notably, during the project, an issue with barcode scanner cameras being installed upside down was identified, which had previously led to misleading test results. This oversight emphasized the necessity for more stringent testing protocols and reinforced the significance of comprehensive quality checks in regulatory processes.
These advantages collectively enhance a more robust governance structure and a more resilient organization, positioning companies to thrive in a complex oversight landscape.
Challenges Faced by Pharmaceutical Compliance Officers
Pharmaceutical oversight officers encounter a multitude of obstacles that significantly impact their ability to ensure adherence to regulations. Key challenges include:
- : The regulatory landscape is in a constant state of evolution, compelling organizations to stay informed about new requirements. Notably, ISO has established across 173 nations, highlighting the complexity of the environment in which conformity officers operate. This extensive array of standards demands a comprehensive understanding of both local and international regulations, making it imperative for to remain vigilant and well-informed.
- : Many regulatory teams find themselves stretched thin, often functioning with limited personnel and budgetary resources. This scarcity can impede their ability to effectively manage regulatory requirements, resulting in heightened risks of non-adherence. Alarmingly, statistics reveal that 98% of organizations maintain a relationship with at least one third-party that has experienced a breach in the past two years. This statistic underscores the urgent need for robust , as the repercussions of non-compliance can be severe, including financial penalties and reputational damage. Engaging with AVS can provide essential support to enhance management processes, particularly for organizations facing resource limitations.
- : The intricate nature of regulations can lead to confusion and misinterpretation, thereby increasing the risk of non-compliance. Compliance officers must navigate a labyrinth of guidelines, necessitating a clear understanding of both local and international regulations. The ethical considerations associated with these regulations further complicate the adherence landscape, as pharmaceutical companies must reconcile conformity with ethical business practices. AVS Life Sciences offers comprehensive GXP regulatory services, including , to assist organizations in effectively navigating these complexities.
- : Cultivating a culture of adherence within an organization can prove particularly challenging, especially in the face of opposition from staff. Overcoming this cultural barrier is crucial to ensuring that adherence is viewed as a collective responsibility rather than a mere obligation. Engaging employees at every level and emphasizing the importance of adherence can help mitigate this resistance.
- : Accurate and thorough documentation is vital for adherence; however, many organizations grapple with efficient practices. This challenge is exacerbated by the increasing volume of data that must oversee, necessitating the implementation of effective data management systems. The complexity of managing this data can overwhelm regulatory teams, further straining their already limited resources.
Addressing these challenges often requires the expertise of regulatory consultants who can provide and targeted strategies. For instance, case studies have illustrated that organizations facing have successfully leveraged , such as those offered by AVS Life Sciences, to enhance their management processes. A notable example is the influence of India and China in global pharmaceutical supply chains, where regulatory challenges are shaped by geopolitical factors and market demands.
By engaging expert advisors, businesses can not only navigate the intricacies of adherence but also adopt sustainable practices that align with legal standards. As Robert Half highlighted, "highly skilled professionals such as compliance officers can be difficult to locate in today’s hiring market," emphasizing the human resource challenges inherent in regulatory management. As the landscape continues to evolve in 2025, the role of oversight officers will be more critical than ever in safeguarding organizational integrity and ensuring adherence to regulatory standards.
For tailored pharmaceutical compliance consulting services, regulatory officers are encouraged to connect with AVS Life Sciences to enhance their management processes.

Future Trends in Pharmaceutical Compliance Consulting
The future of pharmaceutical regulatory consulting is poised for significant transformation, driven by several crucial trends:
- : The anticipated use of digital tools and technologies is set to revolutionize regulatory processes, enhancing data management and operational efficiency. This shift will empower organizations to respond swiftly to regulatory changes and streamline adherence workflows. However, cultural barriers often impede within organizations. Leadership must effectively communicate changes and foster a supportive culture to overcome these challenges. Case studies reveal that addressing cultural issues can expedite digital transformation and improve workflows.
- : AI is expected to become indispensable in predictive analytics for risk management. By harnessing AI, companies can implement proactive measures that significantly reduce regulatory violations and audit findings. According to the PwC Health Research Institute, pharmaceutical firms utilizing predictive analytics for risk management have reported a 40% reduction in violations and a 25% decrease in audit findings, underscoring the advantages of AI in bolstering overall adherence to regulations.
- : As governing bodies intensify scrutiny, pharmaceutical companies must fortify their frameworks to meet elevated expectations. This trend necessitates a more robust approach to , ensuring organizations are well-prepared for rigorous evaluations. Companies are encouraged to establish regulatory intelligence initiatives and adopt a risk-oriented strategy to enhance the value of regulatory analytics, which is essential for and aligns with the demand for flexible governance frameworks. AVS Life Sciences offers expert solutions in , validation, and engineering, assisting firms in navigating these challenges effectively. Additionally, incorporating GXP standards, CAPA processes, and Standard Operating Procedures (SOPs) will further bolster adherence efforts.
- : The integration of sustainability practices into regulatory consulting is gaining momentum, reflecting a broader global commitment to environmental responsibility. Businesses will increasingly need to align their adherence strategies with sustainability objectives, addressing both legal obligations and societal expectations.
- : Efforts to unify regulations across regions are expected to facilitate adherence for multinational enterprises. This trend will enable more seamless operations and reduce the complexities associated with navigating diverse oversight environments.
These trends underscore the necessity for pharmaceutical firms to engage in [pharmaceutical compliance consulting services](https://numberanalytics.com/blog/regulatory-analytics-healthcare-pharma-trends) to remain agile and responsive to the evolving adherence landscape, ensuring they are adequately equipped to confront upcoming challenges. AVS Life Sciences, with its impressive 80% repeat business rate, exemplifies a leader in the market, demonstrating a commitment to excellence and client satisfaction in navigating these transformative trends. Furthermore, understanding the is crucial for regulatory alignment and informed business decision-making, as it plays a pivotal role in data management and adherence to regulations.
To deepen this understanding, we encourage participation in our upcoming webinar, which will thoroughly examine the and its implications for compliance.

Conclusion
In the intricate landscape of the pharmaceutical industry, compliance consulting stands as a fundamental pillar for ensuring safety, efficacy, and public trust. The article underscores the pivotal role of compliance consultants in navigating regulatory frameworks, highlighting their expertise in Good Manufacturing Practices (GMP), Quality System Regulations (QSR), and international standards. With an alarming 98% of organizations encountering compliance breaches recently, the demand for robust compliance frameworks has never been more urgent.
Engaging compliance consultants not only enhances operational efficiency but also protects against costly penalties and reputational harm. Through compelling case studies, such as the successful upgrade of a biotechnology facility by AVS Life Sciences, the transformative impact of expert guidance becomes clear. These consultants streamline processes, implement quality management systems, and provide training that cultivates a culture of compliance, ultimately elevating product quality and patient safety.
As the regulatory landscape continues to shift, the future of pharmaceutical compliance consulting will be influenced by digital transformation, artificial intelligence, and a heightened focus on sustainability. Companies must remain agile, adapting to increased regulatory scrutiny while integrating innovative solutions to bolster compliance frameworks. AVS Life Sciences exemplifies this proactive stance, positioning itself as an industry leader.
In conclusion, the significance of pharmaceutical compliance consulting cannot be overstated. It is essential for organizations aiming to maintain regulatory adherence, enhance operational efficiency, and uphold their commitment to quality and safety. By prioritizing compliance, companies not only mitigate risks but also nurture public trust, ultimately contributing to improved health outcomes for patients globally.
Frequently Asked Questions
What are pharmaceutical compliance consulting services?
Pharmaceutical compliance consulting services are specialized advisory roles that help pharmaceutical firms adhere to legal standards and best practices, focusing on areas such as Good Manufacturing Practices (GMP), Quality System Regulations (QSR), and International Organization for Standardization (ISO) standards.
Why are regulatory consulting services important for pharmaceutical companies?
Regulatory consulting services are essential due to the complex regulatory landscape, helping companies mitigate risks associated with compliance breaches, which can lead to severe fines, legal repercussions, and threats to public health.
What are some key functions of pharmaceutical compliance consulting services?
Key functions include conducting thorough audits, developing oversight programs, and providing training to ensure staff are knowledgeable about legal requirements, particularly in areas like GMP adherence and data integrity.
How can compliance consulting services benefit pharmaceutical companies financially?
Engaging compliance consulting services can lead to significant cost savings; for example, businesses can save an average of $1.25 million in compliance costs by appointing a dedicated compliance leader.
Can you provide an example of a successful case study in pharmaceutical compliance consulting?
AVS Health successfully assisted a biotechnology firm in upgrading their GMP facility from Biosafety Level 1 to Level 2, completing the project on time and within budget while ensuring robust quality assurance procedures.
What are the consequences of non-compliance in the pharmaceutical industry?
Non-compliance can result in substantial fines, legal issues, loss of market access, negative health outcomes for patients, and a decline in public confidence in pharmaceutical products.
How do AVS Life Sciences' services enhance operational outcomes for clients?
AVS Life Sciences' thorough quality management and regulatory adherence solutions improve clients' operations by ensuring compliance, reducing noncompliance rates, and allowing clients to focus on their core mission of developing medications.
What current trends are influencing the demand for compliance consulting services in the pharmaceutical industry?
There is a growing focus on proactive adherence strategies, with many organizations integrating compliance consulting services into their operations to address specific legal challenges and enhance operational efficiency.
How does AVS Life Sciences ensure the effectiveness of its consulting services?
AVS Life Sciences emphasizes thoroughness and professionalism in its consulting approach, which contributes to timely project delivery and positive audit outcomes, positioning clients as leaders in the life sciences industry.
